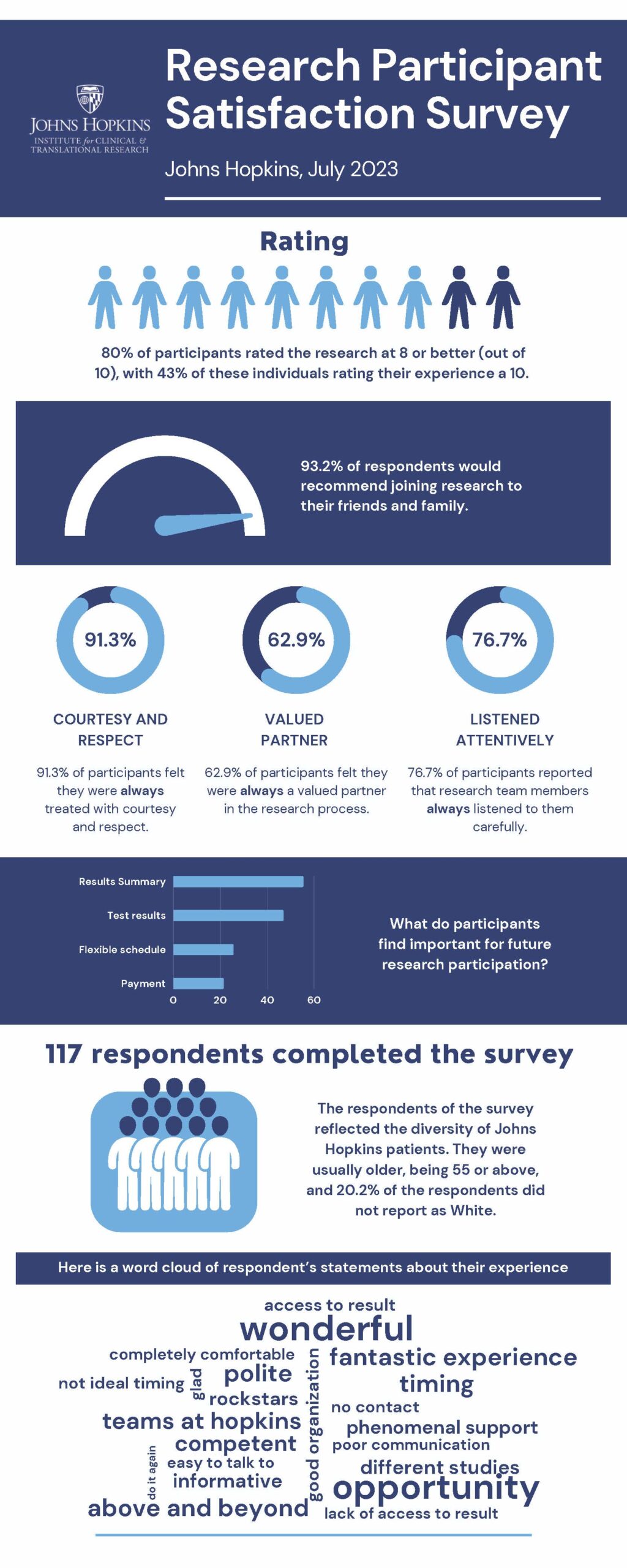
Johns Hopkins Medicine is committed to making the experience of being a research participant as positive as possible. We can only do this if we give research participants the opportunity to rate their experience. Every six months since 2016, the ICTR identifies a random sample of 500 research participants to receive a survey. In the survey, we ask about the informed consent process, interaction with the research team, and how easy it was to complete the study. We pledge to all who completed the survey that the results will be posted for the public and patients to review.
For questions about the survey or if you would like to learn more about the research participant experience, contact our Research Participant Advocate Liz Martinez, RN, BSN, CCRC at 410-614-6323 or [email protected].
Download the July 2023 Results